Android is a trademark of Google LLC.
"Google Play" is a property of Google LLC.
AppStore® online store is a service mark of Apple Inc.
Not sure about your phone model?
Identify your iPhone®† mobile digital device model number
Identify your Samsung®§ mobile device model number
Frequently asked questions
To identify the model number of your device, see below:
- Settings > About Phone
Note: Android™ phone devices (e.g., Samsung Galaxy 10) have multiple model numbers, which indicate details specific to the device. These model numbers are listed above in parenthesis (e.g. SM-G973F). Only Android phone device model numbers listed in the table above are compatible with the MiniMed™ 770G system. The device model number can be found by navigating to the phone’s settings, opening device settings, and then by tapping “About phone.”

To identify the model number of your device, see below:
- Settings > General > About
If your mobile device isn't on the compatible list, you won't be able to use the MiniMed™ Mobile app. However, you can still use the MiniMed™ 780G pump, Simplera Sync sensor, Guardian 4 transmitter and sensor without the app.
If you are using the Instinct sensor, the MiniMed™ Mobile app is required to pair your pump and sensor. In this case, you will need a compatible device to complete the setup and use the sensor.
The App Manager is an accessory that comes with the MiniMed™ Mobile app already installed. It helps you pair your Instinct sensor, made by Abbott, with your MiniMed™ 780G insulin pump. This no-cost accessory is available for you if you have an insurance plan that requires us to include it as part of your sensor order, and/or if you don’t have a smartphone that is compatible with the MiniMed™ Mobile app.
Turning off Automatic Updates on your mobile device may help you avoid unintentionally updating to an operating system that is not confirmed as compatible with the MiniMed™ 780G system mobile apps.
The MiniMed™ 780G system is a medical device and we do extensive testing on all updates. You may experience a delay from when Apple or Android™ releases a new operating system / device model and when the mobile apps are compatible. Before updating your operating system or device, check the compatibility list above.
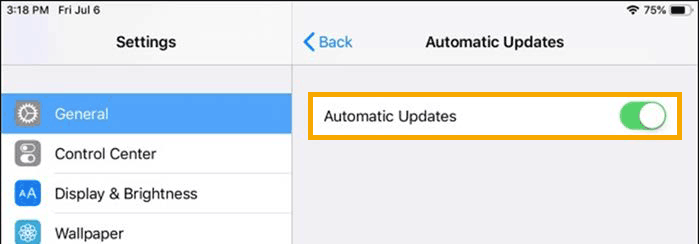
From the settings menu, go to General, go to Software Updates and set Automatic Updates to the “off” position.
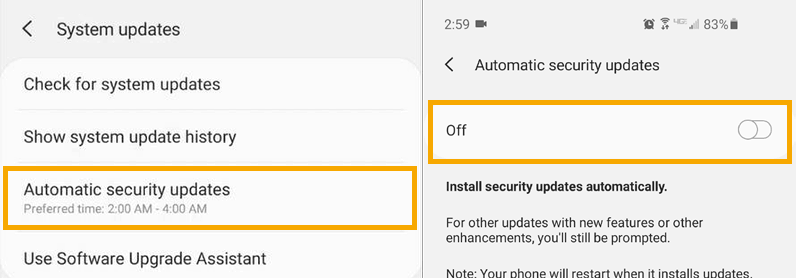
From the System updates menu, select Automatic security updates and turn OFF.
† iPhone® mobile digital device is a registered trademark of Apple Inc.
§ Samsung® is a registered trademark of Samsung.
∆ Smart devices sold separately. For a list of compatible devices, refer to user guide.
The MiniMed™ 780G system is intended for the continuous delivery of basal insulin at selectable rates and the administration of insulin boluses at selectable rates for the management of type 1 diabetes mellitus in persons 7 years of age and older, and of type 2 diabetes mellitus in persons 18 years of age and older requiring insulin. The system is also intended to continuously monitor glucose vales in the fluid under the skin.
The MiniMed™ 780G System includes SmartGuard™ technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values. The system is intended for use with connected sensors, including the Simplera Sync™ and Guardian™ 4 sensors and integrated continuous glucose monitors, including the Instinct sensor, each of which has different wear-time, form factor, insertion site, and other distinguishing characteristics that relate to sensor performance. Consult the appropriate sensor user guide when using the system. Discuss treatment decisions with your HCP.
WARNING: Do not use the SmartGuard™ feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuard™ feature.
WARNING: Do not use the MiniMed™ 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of the MiniMed™ 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuard™ feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a blood glucose (BG) meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMed™ 780G system has not been studied in pregnant women or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including accessories and additional important safety information concerning indications, contraindications, warnings and precautions associated with the system and its components, please consult https://www.medtronicdiabetes.com/important-safety-information and the appropriate user guides at https://www.medtronicdiabetes.com/download-library.
Important Safety Information: MiniMed™ Mobile app
The MiniMed™ Mobile app is intended to provide a secondary display for a compatible MiniMed™ insulin pump system on a suitable consumer electronic device for passive monitoring and to sync data to CareLink™ system. The MiniMed™ Mobile app is not intended to replace the real-time display of Continuous glucose monitoring or insulin pump data on the primary display device (i.e., the insulin pump). All therapy decisions should be based on the primary display device. The MiniMed™ Mobile app is not intended to analyze or modify the continuous glucose monitoring data or insulin pump data that it receives. Nor is it intended to control any function of the connected continuous glucose monitoring system or insulin pump. The MiniMed™ Mobile app is not intended to receive information directly from the sensor or transmitter of a continuous glucose monitoring system.
Important Safety Information: CareLink™ Connect app
The CareLink™ Connect app is intended to provide a secondary display of insulin pump and CGM (Continuous Glucose Monitor) data on a supported mobile device. CareLink™ Connect app is not intended to replace the real-time display of insulin pump or CGM data on the primary display device. All therapy decisions should be based on the primary display device. CareLink™ Connect app is not intended to analyze or modify the insulin pump and CGM data that it receives. Nor is it intended to control any function of the insulin pump or CGM system to which it is connected. CareLink™ Connect app is not intended to receive information directly from the insulin pump or CGM system.
Important Safety Information: Guardian™ Connect app
The Guardian™ Connect system requires a prescription and is indicated for continuous or periodic monitoring of glucose levels in the interstitial fluid under the skin, in patients (14 to 75 years of age) with diabetes mellitus. The system is intended to complement, not replace, information obtained from standard blood glucose monitoring devices, and is not recommended for people who are unwilling or unable to perform a minimum of two meter blood glucose tests per day, or for people who are unable or unwilling to maintain contact with their healthcare professional. The system requires a functioning mobile electronic device with correct settings. If the mobile device is not set up or used correctly, you may not receive sensor glucose information or alerts. For complete details of the system and its components, including warnings, contraindications, and precautions, please consult the user guide at http://www.medtronicdiabetes.com/support/download-library/user-guides and important safety information.