
My Insights
Powered by you.
Personalized for you.
Are you a Time in Range Trooper?
Bolus Boss? Glucose Gladiator?
Find out with a monthly Medtronic email that shares insights like you’ve never seen before, exclusively for people using a MiniMed™ 700G series insulin pump and a connected sensor.
#MiniMedChampion since 2018
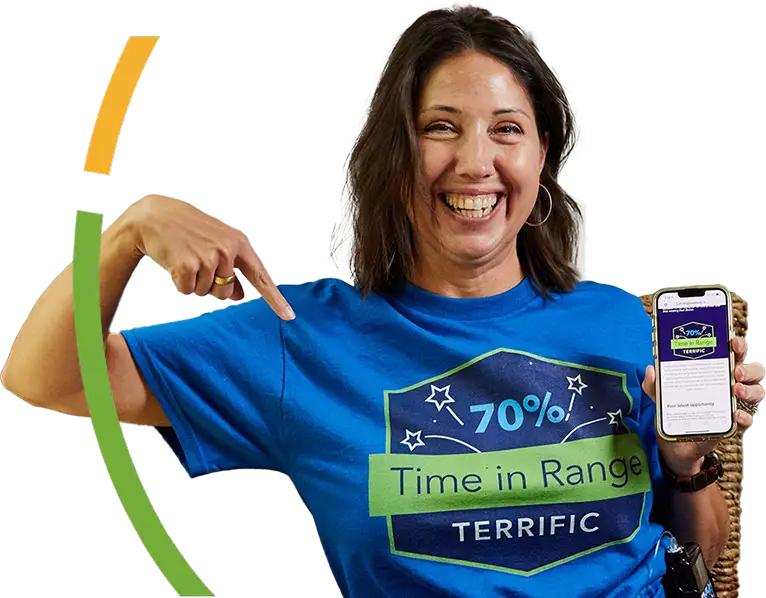
#MiniMedChampion since 2018
My Insights emails include:



What others are saying about My Insights
Get My Insights for your MiniMed™ 700 series system
As easy as 1-2-3
A MiniMed™ 780G or MiniMed™ 770G system + Diabetes.shop account + data uploading to CareLink™ software
Subscribe
Click here to set up a CareLink™ Personal account
During registration, simply check the box that you want to receive the My Insights email.
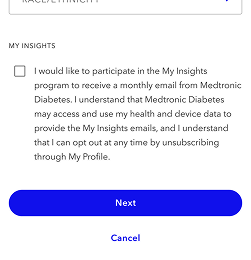
Log in and go to the My Profile section. Scroll to the My Insights section and edit your settings.
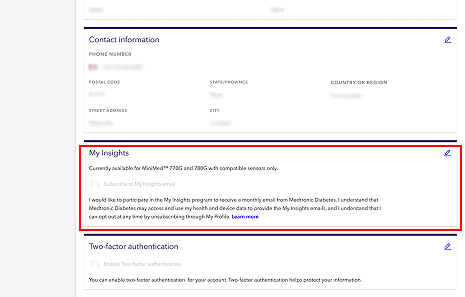
Confirm that your data is uploading
Make sure your MiniMed™ 780G or MiniMed™ 770G device is uploading to CareLink™ Personal every 2 weeks. This can be done automatically through the “Sync to CareLink” daily upload feature on the MiniMed™ Mobile app, or via the blue adapter.
Continue to wear your pump and sensor
Once you’ve given us permission and we have a few weeks of data, expect to get your first email on the next scheduled send. If you wear your system and data continues to upload, then you will receive monthly emails (since tips are related to the pump and sensor).
Frequently asked questions
My Insights are powered by your full system, so you won’t receive insights if you’ve taken a pump or sensor break for longer than 14 days within the last month. We look forward to getting you back on track with your My Insights email.
If that’s not the case, here are some other reasons why your data might not be uploading:
- Check to see if you’re still logged in to the MiniMed™ Mobile app.
- Make sure "Sync to CareLink™" is turned on. Here’s how: Tap on the three lines in the top left corner of the app and select the sync option. If it’s on, you will see a green bar on the next screen. If it’s turned off, you will see a white circle with no bar. Tap to turn on.
- Look at when the last app sync occurred. If it was more than 24 hours ago, tap “Upload Now”.
If you don’t use the app then upload to CareLink™ Personal every 2 weeks using the blue adapter. Still need help? Call 877-379-6242 to speak with a support representative Monday-Friday, 9 a.m. - 6 p.m. CT.
There are a few simple steps to check to make sure you continue to receive emails. You should be able to answer yes to each of these:
- Did you subscribe to the program more than two weeks ago? Please note, once we have the minimum duration of data, depending on what date this is achieved, it could take several more weeks until we send another round of emails.
- Is your data uploading to CareLink™ Personal (via the MiniMed™ Mobile app with automatic sync turned on, or through bi-weekly manual uploads with the blue adapter)?
- Have you worn both your pump and sensor for more than two weeks in the last month?
If you meet these criteria and still need help then please call 877-379-6242 to speak with a support representative Monday-Friday, 9 a.m. - 6 p.m. CT.
Opt in now to earn your first badge, like Melanie, who's a Time in Range Trooper!




Important Safety Information: MiniMed™ 780G System With SmartGuard™ Technology With Guardian™ 4 Sensor
The MiniMed™ 780G system is intended for continuous delivery of basal insulin at selectable rates, and the administration of insulin boluses at selectable amounts for the management of type 1 diabetes mellitus in persons seven years of age and older requiring insulin as well as for the continuous monitoring and trending of glucose levels in the fluid under the skin. The MiniMed™ 780G system includes SmartGuard™ technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values.
The Medtronic MiniMed™ 780G system consists of the following devices: MiniMed™ 780G insulin pump, the Guardian™ 4 transmitter, the Guardian™ 4 sensor, One-press serter, the Accu-Chek™ Guide Link blood glucose meter, and the Accu-Chek™ Guide test strips. The system requires a prescription from a healthcare professional.
The Guardian™ 4 sensor is intended for use with the MiniMed™ 780G system and the Guardian 4 transmitter to monitor glucose levels for the management of diabetes. The sensor is intended for single use and requires a prescription. The Guardian™ 4 sensor is indicated for up to seven days of continuous use.
The Guardian™ 4 sensor is not intended to be used directly to make therapy adjustments while the MiniMed™ 780G is operating in manual mode. All therapy adjustments in manual mode should be based on measurements obtained using a blood glucose meter and not on values provided by the Guardian™ 4 sensor. The Guardian™ 4 sensor has been studied and is approved for use in patients ages 7 years and older and in the arm insertion site only. Do not use the Guardian™ 4 sensor in the abdomen or other body sites including the buttocks, due to unknown or different performance that could result in hypoglycemia or hyperglycemia.
WARNING: Do not use the SmartGuard™ feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuard™ feature.
WARNING: Do not use the MiniMed™ 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of the MiniMed™ 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuard™ feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a BG meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMed™ 780G system has not been studied in pregnant women, persons with type 2 diabetes, or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including product and important safety information such as indications, contraindications, warnings and precautions associated with system and its components, please consult https://www.medtronicdiabetes.com/important-safety-information#minimed-780g and the appropriate user guide at https://www.medtronicdiabetes.com/download-library




